8) Racemising 3-aryl-3-hydroxy-2-oxindoles in a Pickering emulsion under acid catalysis and its application to dynamic kinetic resolution
Moon, Jihoon, Shuji Akai, and Kyohei Kanomata.
Org.Biomol. Chem.
DOI: 10.1039/d6ob00544f.
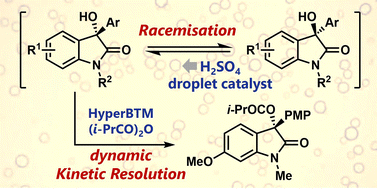
Abstract: 3-Aryl-3-hydroxy-2-oxindoles were racemised under acid-catalysis in a water-in-oil Pickering emulsion comprising toluene and aqueous H2SO4. The developed biphasic methodology overcomes the decomposition issues often encountered during alcohol racemisation and affords high substrate recoveries. Successfully combining racemisation with enantioselective acylation led to the first dynamic kinetic resolution of an oxindole derivative.

7) Site-selective Deuteration of Thioacetals using Self-prepared KOD in D2O
Hyoga Shimizu, Yuto Suganuma, Keisuke Imai, Taro Udagawa, Kyohei Kanomata, Shuji Akai and Yoshinari Sawama*
Asian Journal of Organic Chemistry , 2026; 15:e70443
DOI:10.1002/ajoc.70443
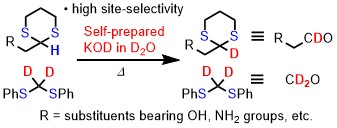
Abstract: Deuterium-incorporated organic compounds are used in various scientific fields. Therefore, the development of direct deuteration (H/D exchange reaction) is highly desired. We developed a simple and cost-effective method for preparing potassium deuteroxide (KOD) in D2O and applied it in the site-selective deuteration of thioacetals as aldehyde equivalents. This approach provided high deuteration contents across a wide range of substrates, bearing hydroxyl, amino, acetal, and other moieties. The obtained deuterated thioacetals were further converted into the corresponding deuterated aldehydes, methylene-bridged diols, and natural products. This deuteration method is a practical and efficient approach for synthesizing deuterated compounds and contributes to the development of deuterated pharmaceuticals and advanced materials.

6) Platinum-group-metal-on-carbon catalyzed dehydration of tertiary alcohols and its application to the one-pot synthesis of aromatics
Shuki Oyama, Yuto Suganuma, Rina Adachi, Shuji Akai and Yoshinari Sawama *
Catal. Sci. Technol.,2026, 16,3536–3541
DOI: 10.1039/d6cy00165c
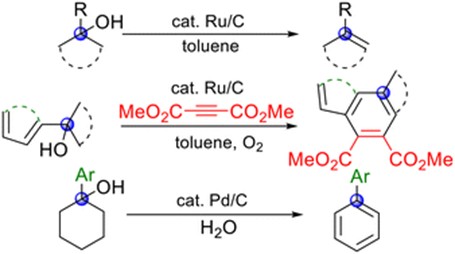
Abstract: Catalytic dehydration of alcohols remains a challenge in organic synthesis, and olefin products are important synthetic precursors because of their high reactivity. Platinum-group-metal-on-carbon catalysts (such as Ru/C and Pd/C) are commercially available and widely utilized as heterogeneous, green, and sustainable catalysts in various organic reactions. However, examples of their application to the catalytic dehydration of alcohols have not been reported. Herein, we demonstrate the Ru/C-catalyzed dehydration of tertiary (tert) alcohols in toluene under thermal conditions. Ru/C also potentially exhibits oxidative activity. Therefore, a tandem reaction involving the Ru/C-catalyzed dehydration of tert-benzylic alcohols, a Diels–Alder reaction, and subsequent oxygen oxidation was developed to afford highly functionalized aromatic products. Additionally, a one-pot synthesis of biaryls from 1-arylcyclohexanols as tert-alcohol substrates was successfully developed via Pd/C-catalyzed dehydration and subsequent dehydrogenation in H2O. These green and sustainable synthetic approaches based on novel heterogeneous dehydration methodologies are valuable for the construction of diverse functional materials.

5) Porcine Liver Esterase-Catalyzed Dynamic Kinetic Resolution of a Lactone-Bridged Biaryl: Impact of Organic Cosolvents on Enantioselectivity
Neha Dhiman, Anke Hummel, Kyohei Kanomata, Yoshinari Sawama, Tohru Taniguchi, Harald Gröger, Shuji Akai
European Journal of Organic Chemistry, 2026; e70473
DOI: 10.1002/ejoc.70473
Open Access
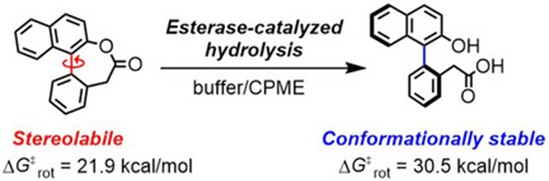
Abstract: A stereolabile lactone-bridged biaryl enables dynamic kinetic resolution to provide a conformationally stable ring-opened biaryl. A significant positive effect of water-immiscible organic cosolvents on the enantioselectivity in a reaction catalyzed by porcine liver esterase has been discovered for the first time.

4) 重水素創薬を拡充する重アルキル化剤
澤間 善成、阪 一穂
和光純薬時報, Vol.94, No.2, 2026年4月

3) 重医薬品開発を指向した位置選択的重水素化法
澤間 善成
有機合成化学協会誌, 2026年1月号, p.50-61

2) Controlled formation of versatile methylated compounds based on ring opening of 4-methyl-1-siloxy-1,4-epoxy-1,4-dihydrobenzene
Takaaki Aijima,‡ Jin Tokunaga,‡ Sota Yoshimura, Yuki Itabashi, Tsunayoshi Takehara, Takeyuki Suzuki, Shuji Akai, Yoshinari Sawama
(‡equally contributed)
RSC Adv. 2026, 16, 15586–15590.
DOI: 10.1039/D6RA01853J
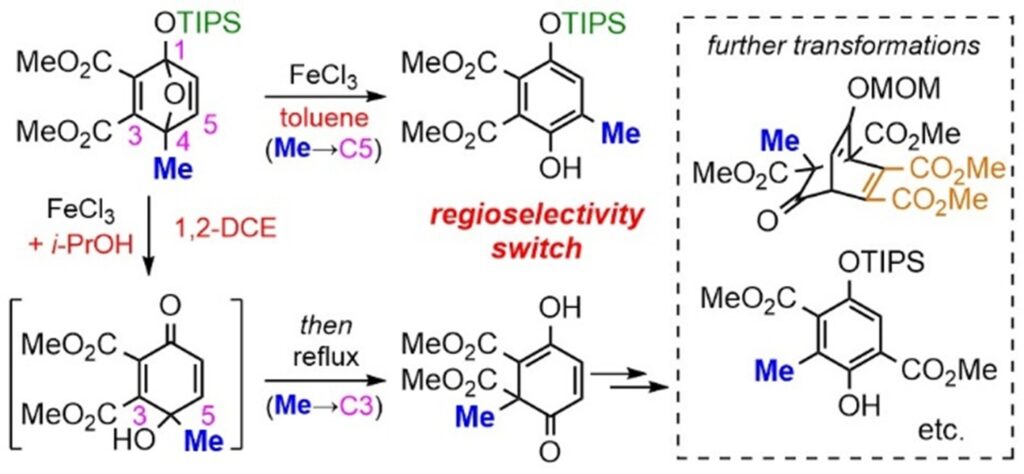
Abstract: We report an FeCl3-catalyzed transformation of 4-methyl-1-siloxy-1,4-epoxy-1,4-dihydrobenzene. Reaction in toluene gave the phenol product, whereas the addition of i-PrOH in 1,2-dichloroethane induced desilylative ring opening to produce 4-hydroxy-4-methyl-2,4-cyclohexadienone, which subsequently underwent a CO2Me-induced regioselective 1,2-methyl shift (C4 to C3) to afford 6-methyl-2,4-cyclohexadienone. This product bears a methyl-substituted quaternary carbon center that is difficult to access by existing methods and serves as a versatile intermediate for further structural elaboration. These results highlight a new mode of skeletal rearrangement and demonstrate regioselective control over competing reaction pathways.

1) Synthetic Studies Toward Rubioncolin B
Takaaki Aijima, Shuji Akai, Yoshinari Sawama
Eur. J. Org. Chem. 2026, 29, e202501103.
DOI: 10.1002/ejoc.202501103
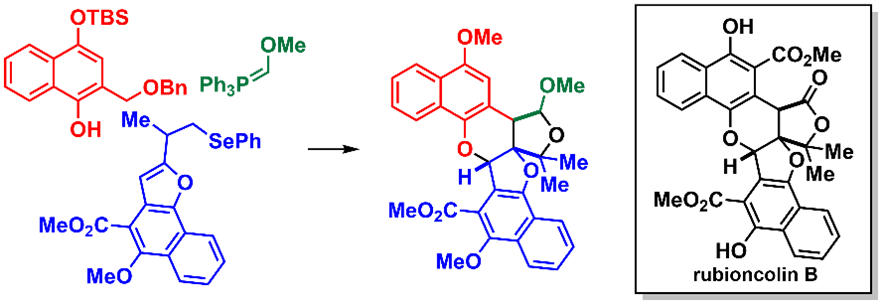
Abstract: Herein, we report our synthetic studies toward rubioncolin B (1), a heptacyclic naphthohydroquinone dimer with antitumor activity. Our strategy is based on the intermolecular [4 + 2]-cycloaddition reaction between an ortho-naphthoquinone methide (o-NQM), which is generated in situ from a 1-naphthol derivative under catalytic Lewis acid conditions, and a naphthofuran derivative, thereby enabling the construction of a hexacyclic compound. We then explored three synthetic approaches for appending the remaining ring structure to the hexacyclic compound. Among them, the sequence involving the introduction of a Wittig reagent and subsequent cyclization proved effective, furnishing a cyclic acetal with the same ring structure as 1. This strategy not only demonstrates that accessing the highly congested architecture of 1 is feasible but also provides a versatile platform for the synthesis of structurally diverse analogs, thereby facilitating subsequent biological investigations, including structure–activity relationship studies.

